Content Menu
● How Activated Carbon Works In Water
>> Key Mechanisms Of Contaminant Removal
● Chlorine, Chloramine And Disinfection By‑Products
>> Taste, Odor And Aesthetic Issues
● Volatile Organic Compounds (VOCs) And Industrial Chemicals
>> Pesticides, Herbicides And Agricultural Residues
● Pharmaceuticals, Micropollutants And PFAS
>> Removal Of PFAS (“Forever Chemicals”)
● Heavy Metals And Inorganic Contaminants
>> Microplastics And Fine Particles
● Factors That Influence Activated Carbon Performance
● Types Of Activated Carbon Water Filters
>> Carbon Block And Point‑Of‑Use Filters
● Typical Applications Of Activated Carbon In Water Treatment
● Conclusion
● FAQs About Activated Carbon And Water Treatment
>> 1. How does activated carbon remove contaminants from water?
>> 2. Which contaminants can activated carbon not remove effectively?
>> 3. How long does activated carbon last in a water filter?
>> 4. Is activated carbon safe for drinking water applications?
>> 5. Why combine activated carbon with other water treatment technologies?
● Citations:
Activated carbon is a highly porous filtration material made from coal, coconut shell, wood, or other carbon‑rich sources that are processed to create an enormous internal surface area. In water treatment, activated carbon is one of the most common and effective technologies for polishing water quality by removing dissolved contaminants that traditional filtration cannot catch.[3][5][6][8][1]
When water flows through a bed or cartridge of activated carbon, contaminants are attracted to and held on the carbon surface, while cleaner water exits the filter. This makes activated carbon essential in municipal water plants, industrial water systems, food and beverage production, and point‑of‑use filters for homes and offices.[9][10][11][6][3]

How Activated Carbon Works In Water
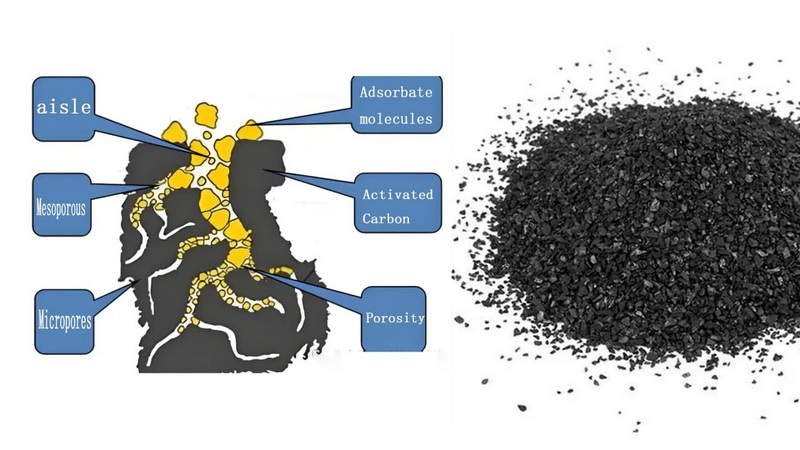
Activated carbon works primarily through adsorption, not absorption. Adsorption means contaminants adhere to the surface of activated carbon pores due to physical and chemical attractions, rather than being absorbed into the bulk of the material.[6][12][3]
A typical activated carbon particle has a specific surface area of around 1,000 m² per gram or even higher, which gives it a huge number of sites to capture molecules from water. In addition, certain activated carbon grades can catalytically convert reactive species like chlorine into less harmful forms, further improving water quality.[5][1][3]
Key Mechanisms Of Contaminant Removal
- Physical adsorption: Non‑polar organic molecules such as VOCs and many industrial chemicals are attracted to the non‑polar activated carbon surface and retained in its pores.[3][6]
- Catalytic reduction: Activated carbon can reduce oxidizing disinfectants like chlorine and chloramine to chloride ions, which are less reactive and less harmful.[1][3]
- Micropore trapping: Very small pores can trap fine particles and some microplastics, improving clarity and safety of drinking water.[13][2]
Chlorine, Chloramine And Disinfection By‑Products
One of the most common uses of activated carbon in water treatment is to remove chlorine and chloramine added by municipalities for disinfection. These disinfectants improve microbiological safety but cause unpleasant taste and odor and can form potentially harmful by‑products.[2][14][1]
Activated carbon very efficiently removes free chlorine and significantly reduces chloramine through catalytic reactions on its surface. As chlorine and chloramine pass through an activated carbon bed, they are converted into chloride ions, so water leaving the filter has greatly reduced disinfectant levels and a much better taste and smell.[1][3]
Taste, Odor And Aesthetic Issues
- Musty or earthy odors from natural organic matter can be adsorbed by activated carbon, resulting in fresher‑tasting water.[15][10]
- Activated carbon also reduces chemical odors and off‑flavors from industrial contaminants, making water more acceptable for drinking and food processing.[2][1]
Because of these effects, activated carbon is widely used in household faucet filters, refrigerator filters, and bottled water production to improve sensory quality.[14][1]
Volatile Organic Compounds (VOCs) And Industrial Chemicals
Activated carbon is particularly effective at removing many volatile organic compounds and other synthetic organic chemicals from water. These include solvents, industrial intermediates, and various organic pollutants that often result from manufacturing, fuel handling, or contaminated groundwater.[7][16][15]
Regulatory and technical sources note that activated carbon can remove or significantly reduce dozens of VOCs and organic chemicals, including trihalomethanes (THMs), haloacetic acids, and other disinfection by‑products. Many water treatment plants and industrial users rely on granular activated carbon (GAC) filters as a polishing step to reduce these organic contaminants to very low levels.[11][15][6][1]
Pesticides, Herbicides And Agricultural Residues
- Activated carbon is widely used to adsorb pesticides and herbicides that reach surface water or groundwater through agricultural runoff.[4][15]
- Compounds such as many chlorinated pesticides and other persistent organic pollutants can be captured by activated carbon's micro‑ and mesopores.[15][4]
This makes activated carbon critical for protecting drinking water sources in agricultural regions and for treating process water in food and beverage industries.[10][4]

Pharmaceuticals, Micropollutants And PFAS
Many modern water quality concerns involve “micropollutants” or “contaminants of emerging concern,” such as pharmaceuticals, personal care products, and other trace organics that traditional treatment does not fully remove. Activated carbon has proven highly effective in adsorbing many of these small and persistent molecules.[17][12][9]
Studies and industrial practice show that activated carbon can remove or significantly reduce various pharmaceuticals, including over‑the‑counter drugs and prescription medicines, from water. Specialized grades of activated carbon are now used in advanced wastewater treatment and drinking water plants to target these emerging contaminants.[9][4][17]
Removal Of PFAS (“Forever Chemicals”)
- PFAS (per‑ and polyfluoroalkyl substances) are a group of highly persistent chemicals used in coatings, firefighting foams, and many industrial applications.[18][4]
- Certain high‑performance activated carbon products are applied to capture PFAS from drinking water and industrial effluents, helping utilities comply with increasingly strict regulations.[18][4]
While activated carbon may not remove all PFAS equally, it remains one of the key technologies for reducing PFAS levels in many water treatment systems.[4][18]
Heavy Metals And Inorganic Contaminants
Activated carbon is primarily designed for organic compounds, but it can also remove or reduce certain inorganic contaminants under the right conditions. For example, some activated carbon filters can reduce levels of metals such as lead, mercury, and copper, especially when these metals are associated with organic complexes.[13][15][2]
However, activated carbon is not equally effective for all inorganic ions. Common anions such as fluoride, nitrate, and hardness ions (calcium and magnesium) are only weakly removed by standard activated carbon and normally require other technologies like ion exchange or reverse osmosis.[19][7][15]
Microplastics And Fine Particles
- Block and finely structured activated carbon filters can capture microplastics down to about 1–2 microns, significantly reducing these particles in finished water.[13][2]
- In many systems, activated carbon is preceded or followed by sediment filters to optimize removal of suspended solids while the activated carbon focuses on dissolved contaminants.[16][2]
This combination helps protect downstream equipment and improves clarity, color, and overall water quality.[10][11]
Factors That Influence Activated Carbon Performance
The effectiveness of activated carbon at removing contaminants from water depends on several key parameters. Understanding these factors is essential when designing industrial activated carbon systems or selecting point‑of‑use filters.[8][11][6][3]
Important factors include:
- Carbon type and raw material: Coconut‑shell, coal‑based, and wood‑based activated carbon have different pore structures that favor different types of contaminants.[11][3]
- Surface area and porosity: Higher surface area and a suitable distribution of micro‑, meso‑, and macropores allow activated carbon to adsorb a broader range of molecules.[12][3]
- Contact time and flow rate: Longer contact time between water and activated carbon typically increases removal efficiency, while very high flow rates reduce performance.[16][3]
- Water chemistry: pH, temperature, and the presence of competing organics or dissolved solids can influence how well specific contaminants are adsorbed.[6][12]
Proper system design considers these variables so that activated carbon can reliably meet industrial, municipal, or household water quality targets.[19][11]
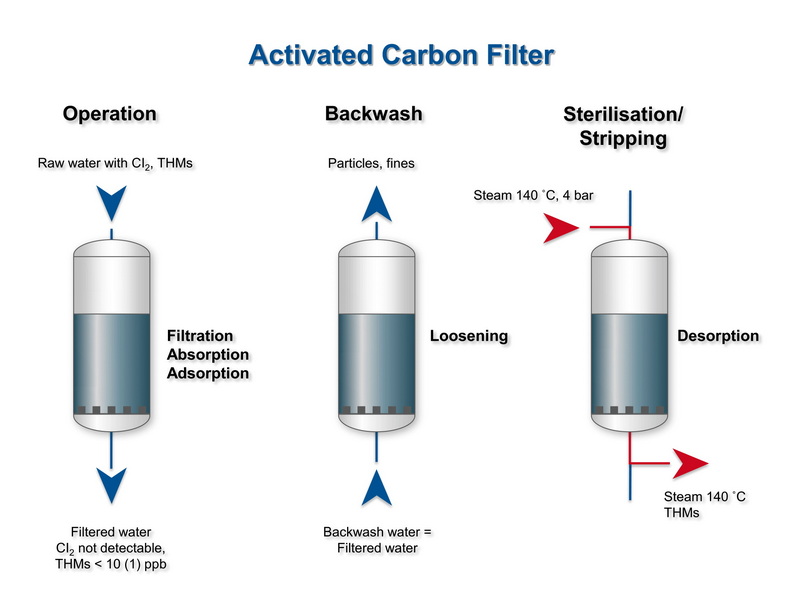
Types Of Activated Carbon Water Filters
There are several common configurations of activated carbon for water purification, each suited to different applications and scales. The main types include granular activated carbon beds, powdered activated carbon dosing, and carbon block filters.[14][3][6]
Granular activated carbon (GAC) filters hold relatively large carbon granules in columns or pressure vessels, allowing continuous treatment of large water flows such as in municipal plants or industrial processes. Powdered activated carbon (PAC) is dosed directly into water, typically in treatment works, and later removed with other sludge during clarification steps.[8][11][6][16]
Carbon Block And Point‑Of‑Use Filters
- Carbon block filters compress fine activated carbon powder into a solid block with controlled porosity, commonly used in under‑sink, countertop, and refrigerator filters.[14][13]
- Many residential filters combine activated carbon with sediment filtration, ion exchange resins, or membranes to broaden contaminant removal capabilities.[13][14]
These designs maximize contact between water and activated carbon, improving adsorption of chlorine, VOCs, and many organic pollutants in domestic drinking water.[10][1]
Typical Applications Of Activated Carbon In Water Treatment
Because activated carbon removes so many dissolved contaminants, it is integrated into numerous water and process treatment systems worldwide. Its flexibility and regenerability also support cost‑effective long‑term operation in demanding industrial environments.[12][9][11][16]
Key application areas include:
- Municipal drinking water treatment: Activated carbon is used as a polishing step to remove taste‑ and odor‑causing compounds, disinfection by‑products, pesticides, and traces of pharmaceuticals before distribution.[9][6]
- Industrial process water: Electronics, chemical, food, and beverage industries use activated carbon to achieve high‑purity water by removing organics and residual chlorine that could damage equipment or affect product quality.[11][10]
- Wastewater and reuse: Advanced activated carbon systems help remove persistent organic pollutants and micropollutants from wastewater before discharge or reuse, protecting the environment and downstream users.[17][9]
In many of these systems, activated carbon functions alongside other technologies such as ion exchange, ultrafiltration, and reverse osmosis to deliver complete water treatment solutions.[19][1]
Conclusion
Activated carbon is one of the most versatile and effective materials for removing contaminants from water, especially chlorine, chloramine, VOCs, pesticides, pharmaceuticals, many micropollutants, PFAS, and taste‑ and odor‑causing compounds. By leveraging physical adsorption, catalytic reactions, and finely engineered pore structures, activated carbon can dramatically improve the safety, taste, and overall quality of drinking water and process water across municipal, industrial, and household systems. For global industrial users who require reliable, high‑performance water treatment, customized activated carbon solutions provide a practical and scalable way to meet strict water quality requirements.[12][3][4][1][9][10][11]
FAQs About Activated Carbon And Water Treatment
1. How does activated carbon remove contaminants from water?
Activated carbon removes contaminants mainly by adsorption, binding dissolved molecules onto its large internal surface area as water passes through its pores. In addition, activated carbon can catalytically reduce disinfectants like chlorine and chloramine, converting them into less reactive chloride ions.[3][6][1]
2. Which contaminants can activated carbon not remove effectively?
Standard activated carbon is not very effective at removing common dissolved salts such as fluoride, nitrate, and hardness ions (calcium and magnesium), and it cannot reliably remove all heavy metals. For these contaminants, technologies such as reverse osmosis, ion exchange, or specialized media are usually required in addition to activated carbon.[15][19]
3. How long does activated carbon last in a water filter?
The service life of activated carbon depends on water quality, contaminant loading, flow rate, and filter size, so there is no universal replacement interval. In household systems, activated carbon cartridges are often replaced every few months, while industrial granular activated carbon beds may operate for many months or years before regeneration or replacement is needed.[8][3][11]
4. Is activated carbon safe for drinking water applications?
Activated carbon produced and certified for drinking water use is considered safe and is widely used in municipal and residential systems worldwide. To ensure safety and performance, users should select activated carbon that meets relevant standards and replace or regenerate the activated carbon as recommended by the manufacturer.[14][1][11][8]
5. Why combine activated carbon with other water treatment technologies?
Activated carbon is excellent for removing many organic contaminants, chlorine, and taste‑ and odor‑causing compounds, but it does not address every possible water quality issue. Combining activated carbon with technologies such as sediment filtration, ion exchange, ultrafiltration, or reverse osmosis allows systems to target suspended solids, dissolved salts, microorganisms, and other contaminants that activated carbon alone cannot fully remove.[7][15][19][1]
Citations:
[1](https://www.springwellwater.com/activated-carbon-filters-remove/)
[2](https://ionexchangeglobal.com/how-an-activated-carbon-filter-cleans-water/)
[3](https://www.watertreatmentguide.com/activated_carbon_filtration.htm)
[4](https://puragen.com/uk/insights/how-does-activated-carbon-filter-water/)
[5](https://www.watertechonline.com/wastewater/article/15549902/the-basics-of-activated-carbon-adsorption)
[6](https://cropaia.com/blog/activated-carbon-in-water-treatment/)
[7](https://espwaterproducts.com/pages/what-do-carbon-filters-remove-from-water)
[8](https://19january2021snapshot.epa.gov/sites/static/files/2015-04/documents/a_citizens_guide_to_activated_carbon_treatment.pdf)
[9](https://en.wikipedia.org/wiki/Activated_carbon)
[10](https://streampeak.com.sg/moisture-absorbers/improve-water-quality-with-activated-carbon-filtration/)
[11](https://generalcarbon.com/understanding-granular-activated-carbon-for-water-treatment/)
[12](https://pubs.acs.org/doi/10.1021/acssusresmgt.4c00414)
[13](https://tappwater.co/blogs/blog/what-activated-carbon-filters-remove)
[14](https://www.freshwatersystems.com/blogs/blog/activated-carbon-filters-101)
[15](https://extension.purdue.edu/extmedia/WQ/WQ-13.html)
[16](https://danamark.com/resources/adsorption-water-filtration/)
[17](https://www.sciencedirect.com/science/article/pii/S2214714424015459)
[18](https://www.saur.com/en/type-solutions/activated-carbon/)
[19](https://www.apecwater.com/blogs/water-quality/quality-water-filtration-method-carbon)
[20](https://www.sciencedirect.com/science/article/pii/S1944398624025426)
















