
Content Menu
● What Is Activated Carbon?
● Chemistry of Ammonia in Water
● Can Standard Activated Carbon Remove Ammonia?
● Modified Activated Carbon for Ammonia Removal
● Activated Carbon, Zeolite, and Hybrid Media
● How Activated Carbon Interacts with Ammonia
● Practical Water Treatment Scenarios
● When to Use Activated Carbon for Ammonia
● Advantages and Limitations of Activated Carbon for Ammonia
● Recommendations for System Design
● Conclusion
● FAQ About Activated Carbon and Ammonia (H3)
>> (1) Does activated carbon remove ammonia from water?
>> (2) Is zeolite better than activated carbon for ammonia?
>> (3) Can powdered activated carbon be used to treat high‑ammonia water?
>> (4) How does pH affect ammonia removal by activated carbon?
>> (5) When should modified activated carbon be considered for ammonia?
● Citations:
Activated carbon can remove a limited amount of ammonia from water, but standard activated carbon is not a highly efficient ammonia adsorbent compared with ion-exchange media such as zeolite or specially modified activated carbon. In practice, engineers often combine activated carbon with zeolite or use modified nano-activated carbon when high ammonia removal is required in drinking water or wastewater treatment.[1][2][3][4]

What Is Activated Carbon?
Activated carbon is a highly porous carbon material with an enormous internal surface area that adsorbs a wide range of organic contaminants, taste and odor compounds, and some inorganic pollutants from water. Activated carbon can be produced from coal, coconut shell, wood, or other carbon‑rich raw materials through carbonization and activation, creating a network of micro‑, meso‑, and macropores.[5][3][6]
Because of this micro‑porous structure, activated carbon is widely used in water treatment, air and gas purification, and many industrial processes such as food and beverage, chemical, and pharmaceutical production. Different forms, such as powdered activated carbon (PAC) and granular activated carbon (GAC), can be selected depending on whether the process is batch dosing, continuous filtration, or advanced polishing.[2][5][1]
Chemistry of Ammonia in Water
To understand whether activated carbon removes ammonia from water, it is important to consider how ammonia behaves in aqueous solution. In water, ammonia exists mainly as un‑ionized ammonia (NH₃) and ammonium ion (NH₄⁺), with the ratio depending strongly on pH and temperature. At higher pH values, more of the total ammonia nitrogen is present as dissolved NH₃, while at lower pH values ammonium ion dominates, which affects which treatment technologies are most effective.[3][5][2]
Ammonia and ammonium are small, highly soluble species, so they do not adsorb onto standard activated carbon surfaces as easily as many hydrophobic organic molecules. That is why water utilities and aquaculture systems often rely on ion‑exchange materials like zeolites, biological nitrification, or membrane processes for strong ammonia control, sometimes using activated carbon only as a complementary polishing step.[7][1][2][3]
Can Standard Activated Carbon Remove Ammonia?
Research shows that conventional activated carbon can adsorb some ammonium nitrogen from water, but the capacity is usually moderate and strongly influenced by pH, dosage, and contact time. In one study, activated carbon prepared from waste tyres achieved around 55–69% removal of ammonia nitrogen under optimized conditions (pH around 9, proper dosage, and 90–150 minutes of contact time). However, the removal efficiency started to decline after longer contact times as adsorption sites became saturated and ionic equilibrium between water and activated carbon was reached.[5]
Other works comparing activated carbon with zeolite in freshwater systems found that activated carbon could temporarily improve water quality and contribute to ammonia reduction, but ammonia levels tended to rebound as the adsorption capacity decreased over time. Overall, standard activated carbon is not considered a primary ammonia removal medium, but it can provide some short‑term or partial ammonia reduction when designed correctly.[1][3]
Modified Activated Carbon for Ammonia Removal
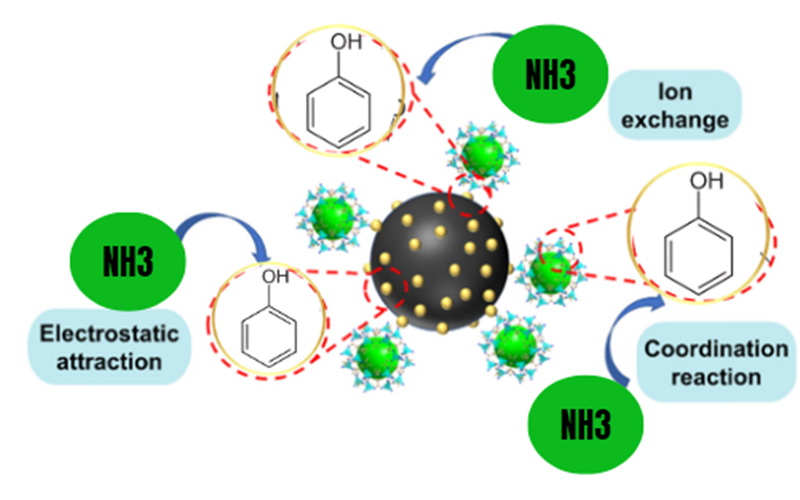
When engineers specifically want activated carbon to remove ammonia more efficiently, they often modify the activated carbon surface to introduce functional groups or nano‑scale features. Laboratory studies show that adding acidic oxygen surface groups to activated carbon significantly enhances ammonia adsorption, especially through interactions between ammonia species and Brønsted and Lewis acid sites on the carbon surface. Under dry or moist air conditions, such surface‑modified activated carbon demonstrated much higher ammonia adsorption capacity than the unmodified material.[4][8][7]
Recent work on nano‑activated carbon has developed highly efficient materials for separating ammonia from water, using tailored surface chemistry and pore structures to increase capacity and improve adsorption kinetics. Such nano‑structured or chemically modified activated carbon products are usually more specialized and targeted for demanding industrial applications where high ammonia loads or strict discharge limits apply.[6][4]
Activated Carbon, Zeolite, and Hybrid Media
Comparative studies often evaluate activated carbon and zeolite side by side for ammonia removal in water. Zeolite, especially certain natural or synthetic types like mordenite and clinoptilolite, can remove ammonium efficiently via ion exchange, which is a different mechanism from the mainly adsorption‑driven process on activated carbon. In high‑ammonia waters, zeolite has been shown to remove more than two‑thirds of ammonia in typical coagulation processes while also reducing precursors of disinfection by‑products.[2][3][1]
Interestingly, several studies highlight that combining activated carbon and zeolite in a hybrid filter or composite media can yield superior performance. One freshwater filter test reported that a hybrid mixture of activated carbon and zeolite reduced ammonia to low levels while maintaining suitable nitrite, nitrate, and pH for fish, with performance improving as the share of hybrid media increased. A review of landfill leachate treatment also noted that composite media of activated carbon and zeolite achieved the best adsorptive ammonia removal, with reported capacities above 24 mg/g.[3][1]
How Activated Carbon Interacts with Ammonia
From a mechanistic point of view, ammonia removal by activated carbon can involve several pathways depending on the aqueous environment and the surface chemistry of the activated carbon. In samples with relatively low concentrations of oxygen surface groups, moisture can promote ammonia removal because ammonia dissolves into water films and then interacts with the carbon surface, improving overall adsorption capacity. In activated carbon samples rich in acidic functional groups, ammonia forms ammonium species that interact with Brønsted acid sites or coordinates with Lewis acid centers on the carbon matrix.[8][7]
Dynamic breakthrough experiments in fixed beds show that operating conditions such as temperature, contact time, and influent ammonia concentration significantly influence the shape of the breakthrough curve and the total adsorption capacity of activated carbon for ammonia. Generally, lower temperatures and properly sized activated carbon beds with sufficient mass of media allow longer breakthrough times before ammonia appears in the effluent.[9][8]
Practical Water Treatment Scenarios
In real water treatment plants, powdered activated carbon and granular activated carbon are rarely used alone when high ammonia removal is required. Instead, activated carbon is typically combined with biological treatment, nitrification, ion‑exchange media, or advanced oxidation processes, with activated carbon focusing on removing organic contaminants, taste and odor compounds, and disinfection‑by‑product precursors. For example, one drinking water study evaluated mordenite zeolite at lime softening and alum coagulation steps, while powdered activated carbon was added to remove specific N‑nitrosamine precursors that zeolite did not capture efficiently.[2][3]
In aquaculture and aquarium systems, activated carbon cartridges may improve clarity and reduce organic pollutants, but zeolite or biological filters are usually recommended to manage ammonia and nitrogen compounds over the long term. In industrial wastewater and landfill leachate, the combination of zeolite and activated carbon – sometimes including “activated zeolite” or composite media – can offer both ammonia reduction and removal of other dissolved organic compounds.[10][11][1][3]
When to Use Activated Carbon for Ammonia
Activated carbon is helpful for ammonia control when the primary goal is to improve overall water quality and remove multiple contaminant classes simultaneously, rather than to target ammonia alone. In moderately contaminated waters where ammonia is present together with organic pollutants and taste and odor compounds, a properly sized activated carbon system can provide partial ammonia removal while delivering strong performance on organic contaminants.[5][1][2]
Activated carbon becomes especially attractive when used in hybrid systems or when modified for higher ammonia selectivity. For example, nano‑activated carbon or impregnated activated carbon can be deployed where space is limited or where operators wish to retrofit existing carbon systems to handle ammonia or related nitrogen species more efficiently.[4][6][3]
Advantages and Limitations of Activated Carbon for Ammonia
Using activated carbon for ammonia removal offers several advantages, but also clear limitations that must be considered in design and operation.[1][3][5]
- Activated carbon advantages for ammonia:
- Provides broad adsorption of organics, odors, and by‑product precursors while also delivering some ammonia reduction.[5][1]
- Can be integrated in existing GAC filters or PAC dosing points without major redesign, which is attractive for retrofits.[3][2]
- Hybrid media and modified activated carbon can significantly increase ammonia removal capacity compared with standard grades.[7][4][3]
- Activated carbon limitations for ammonia:
- Standard activated carbon has relatively low selectivity and capacity for ammonium compared with zeolite or dedicated ion‑exchange resins.[1][2][3]
- Ammonia removal can be temporary, with breakthrough occurring as adsorption sites saturate and equilibrium is reached, causing ammonia levels to rise again.[5][1]
- Performance is sensitive to pH, temperature, and competition from other ions or organics, which complicates prediction of long‑term ammonia removal.[8][7][5]
Recommendations for System Design
For engineers and plant operators evaluating whether activated carbon can remove ammonia from water in their system, several design recommendations can improve results.[2][1][5]
- Evaluate the total ammonia nitrogen load, competing contaminants, and temperature, then decide whether activated carbon alone is sufficient or if hybrid media with zeolite or ion‑exchange resins is required.[3][1][2]
- Consider powdered activated carbon dosing as a flexible, short‑term option when episodic ammonia and organic spikes occur, while using zeolite or biological treatment for base‑load ammonia control.[1][2]
- For continuous filtration applications, design granular activated carbon beds with adequate depth, contact time, and mass of media, and monitor breakthrough curves to schedule media replacement or regeneration before ammonia levels exceed limits.[9][8][5]
- Where high performance is needed, investigate modified or nano‑activated carbon products with tailored surface chemistry for higher ammonia capacity, or composite media that combine activated carbon with zeolite or other sorbents.[6][4][3]
Switching between or combining activated carbon types (coal‑based, coconut‑shell, wood‑based) and forms (PAC, GAC, pellets) also allows customization of pore structure, kinetics, and service life for specific ammonia‑containing water streams.[6][5]
Conclusion
Standard activated carbon can remove some ammonia from water, especially under favorable pH, dosage, and contact time conditions, but it is not the most powerful technology for ammonia control by itself. Zeolite and other ion‑exchange media typically provide higher selectivity and capacity for ammonium, while activated carbon excels at removing organic contaminants, tastes, odors, and disinfection‑by‑product precursors. The most effective strategies for ammonia‑rich waters usually combine activated carbon with zeolite, biological treatment, or modified nano‑activated carbon, allowing simultaneous control of ammonia and other pollutants in a robust, industrially scalable system.[4][6][5][1][2][3]

FAQ About Activated Carbon and Ammonia (H3)
(1) Does activated carbon remove ammonia from water?
Standard activated carbon can remove a limited fraction of ammonia or ammonium from water, but its capacity is generally modest and often temporary as the adsorption sites become saturated. For applications where strict ammonia limits must be met, activated carbon is usually combined with zeolite, biological nitrification, or other ion‑exchange media to achieve reliable long‑term ammonia control.[5][1][2][3]
(2) Is zeolite better than activated carbon for ammonia?
Zeolite is typically more effective than standard activated carbon for removing ammonium from water because it works via ion exchange with a high affinity for ammonium ions. Studies in drinking water treatment, freshwater aquaculture, and landfill leachate show that zeolite can remove more than two‑thirds of ammonia and maintain low ammonia levels over time, while activated carbon alone often shows more temporary effects.[1][2][3]
(3) Can powdered activated carbon be used to treat high‑ammonia water?
Powdered activated carbon can be dosed to high‑ammonia water to provide some ammonia reduction while also removing organic pollutants and by‑product precursors, but it is not usually the sole solution for very high ammonia. A more robust configuration uses powdered activated carbon in combination with zeolite or coagulation steps, where zeolite handles most ammonia removal and powdered activated carbon targets specific organic contaminants that zeolite does not capture.[2][3][5]
(4) How does pH affect ammonia removal by activated carbon?
pH has a strong effect because it controls the balance between un‑ionized ammonia (NH₃) and ammonium ion (NH₄⁺), which interact differently with activated carbon surfaces. Experiments with activated carbon derived from waste tyres showed that ammonia removal was highest around pH 9, while surface‑modified activated carbons with acidic functional groups can adsorb ammonia effectively through acid–base interactions across a range of pH values.[7][5][2]
(5) When should modified activated carbon be considered for ammonia?
Modified or nano‑activated carbon should be considered when space is limited, ammonia levels are high, or when there is a need to handle both ammonia and complex organic contaminants in one compact adsorption step. Research on nano‑activated carbon and surface‑oxidized activated carbon shows that carefully tailored surface chemistry can significantly enhance ammonia adsorption capacity compared with unmodified carbon, making such materials suitable for advanced industrial or environmental applications.[8][7][4][6]
Citations:
[1](https://www.sciencedirect.com/science/article/abs/pii/S221334371930346X)
[2](https://pubmed.ncbi.nlm.nih.gov/29478664/)
[3](https://iwaponline.com/wst/article/84/12/3425/84936/A-review-on-the-application-of-nanoporous-zeolite)
[4](https://pubmed.ncbi.nlm.nih.gov/39121956/)
[5](https://iwaponline.com/wpt/article/18/6/1479/95196/Removal-of-ammonia-nitrogen-from-wastewater-using)
[6](https://allcarbontech.com/activated-carbon-ammonia-removal/)
[7](https://pubs.acs.org/doi/10.1021/es203093v)
[8](https://pmc.ncbi.nlm.nih.gov/articles/PMC8836875/)
[9](https://journals.sagepub.com/doi/pdf/10.1260/026361702321705285)
[10](https://www.aquariacentral.com/forums/threads/carbon-remove-ammonia.83961/)
[11](https://www.walshmedicalmedia.com/open-access/the-applicability-of-activated-carbon-natural-zeolites-and-probiotics-emand-its-effects-on-ammonia-removal-efficiency-and-fry-perf-2155-9546-1000459.pdf)
[12](https://www.perplexity.ai/search/dd6d0a7d-e710-44a9-9f3f-70f59e0da6db)
[13](https://www.sciencedirect.com/science/article/abs/pii/S004565352401943X)
[14](https://generalcarbon.com/activated-carbon/gc-ipa/)
[15](https://pubmed.ncbi.nlm.nih.gov/22049916/)
[16](https://services.jacobi.net/ammonia-removal-from-air/)
[17](https://pubs.acs.org/doi/abs/10.1021/acs.energyfuels.5b00653)
[18](https://www.sciencedirect.com/science/article/pii/S1383586623003623)
[19](https://www.zeolite-products.com/wp-content/uploads/Zeolite-Activated-Carbon.pdf)
[20](https://mp.watereurope.eu/media/uploads/2024/11/08/xue-et-al-2018.pdf)
[21](https://pubs.acs.org/doi/abs/10.1021/es401276r)
















