Content Menu
● What Is Activated Carbon
● Key Types of Activated Carbon for Water
● Mechanisms: How Activated Carbon Removes Contaminants
>> Adsorption on Activated Carbon
>> Catalytic Reduction on Activated Carbon
● What Activated Carbon Removes from Water
● Factors That Control Activated Carbon Performance
● Advantages of Activated Carbon in Water Purification
● Limitations of Activated Carbon in Water Treatment
● Practical Applications of Activated Carbon Water Purification
● Conclusion
● FAQ About Activated Carbon in Water Purification
>> (1) How does activated carbon adsorb contaminants from water?
>> (2) What types of contaminants does activated carbon remove best?
>> (3) What does activated carbon not remove from water?
>> (4) How long does activated carbon last in water filters?
>> (5) What is the difference between granular and powdered activated carbon in water treatment?
● Citations:
How does activated carbon work in water purification? Activated carbon works mainly through adsorption on its extremely porous internal surface, capturing dissolved contaminants as water flows through a packed bed or mixes with fine particles.[1][2]
Activated carbon is a specially processed form of carbon with a huge internal surface area and a network of pores that make it highly effective for removing tastes, odors, and many dissolved pollutants from water. In modern water purification, activated carbon is one of the most widely used adsorbents in municipal plants, industrial systems, and household filters.[3][4][1]

What Is Activated Carbon
Activated carbon is a highly porous carbon material produced from coal, coconut shell, wood, or other carbon‑rich feedstocks and then “activated” to create a very large internal surface area. One gram of activated carbon can have a surface area of thousands of square meters, providing enormous capacity for adsorption of dissolved contaminants from water.[2][6][1]
The internal pore system of activated carbon is tridisperse, typically including macropores, transitional pores, and micropores that connect from the particle exterior into the inner structure. Micropores usually contribute at least 90% of the total internal surface area, making them particularly important for capturing small water contaminants.[1]
- Micropores (≈0.8–2 nm effective radius) are mainly responsible for adsorbing small organic molecules and many trace pollutants.[7][1]
- Transitional (meso) pores help transport molecules from macropores to micropores and contribute to adsorption of somewhat larger compounds.[1]
- Macropores act as transport channels, allowing water and contaminants to enter the particle and then diffuse into smaller pores where adsorption is strongest.[1]
Key Types of Activated Carbon for Water
For water purification, the two main forms of activated carbon are granular activated carbon (GAC) and powdered activated carbon (PAC). Both types use the same basic adsorption mechanism but are applied differently in system design and operation.[4][3][5]
Granular activated carbon consists of relatively large particles used in fixed beds, filter columns, or cartridges, allowing continuous flow and often thermal reactivation and reuse. Powdered activated carbon is a fine powder that is dosed directly into water or process tanks for short‑term or batch treatment and then removed with sedimentation or filtration.[8][9][3][5]
- GAC is commonly used in municipal drinking water plants, industrial water polishing, and household GAC filters for long‑term, continuous service.[9][10]
- PAC is often used for seasonal taste‑and‑odor events, emergency pollution incidents, and industrial process water when flexible dosing is needed.[3][4]
Mechanisms: How Activated Carbon Removes Contaminants
The primary mechanism by which activated carbon works in water purification is adsorption, a surface‑based process in which dissolved molecules accumulate on the carbon surface. In addition, catalytic reduction on activated carbon can transform certain species such as chlorine and chloramine, enhancing overall water quality.[11][4][3]
Adsorption on Activated Carbon
Carbon filtering operates through adsorption, where pollutants in water migrate into the pore structure and are held by attractive forces on the carbon surface. The driving force for adsorption is mainly weak intermolecular interactions (van der Waals forces) combined with the large surface area and pore geometry of activated carbon.[2][7][3]
As water passes through the bed of activated carbon or over PAC particles:
- Dissolved molecules move from the bulk water into the pores by diffusion, with macropores leading to meso‑ and micropores.[3][1]
- Contaminants accumulate in areas of the pore network with the strongest attractive forces, often in narrow micropores.[11][7]
- The balance between the attraction of the carbon surface and the forces holding the contaminants dissolved in water determines how strongly they are adsorbed.[4][11]
Catalytic Reduction on Activated Carbon
For some disinfectants and residual oxidants, activated carbon acts not just as an adsorbent but also as a catalyst. Activated catalytic carbon can chemically convert chlorine into chloride and also help reduce other reactive species, improving taste and reducing byproduct formation.[12][4][11]
In practice, activated carbon:
- Adsorbs many organic compounds and disinfection by‑products directly onto its surface.[10][11]
- Promotes redox reactions that transform free chlorine and, in some cases, chloramines, reducing their concentration and associated odor and taste.[12][4]
What Activated Carbon Removes from Water
Activated carbon removes a broad spectrum of organic and some inorganic contaminants from water, especially those associated with taste, odor, and chemical pollution. The exact removal profile depends on the type of activated carbon, pore size distribution, dosage, contact time, and water chemistry.[10][4][3]
Common contaminant groups removed by activated carbon include:
- Taste and odor compounds, such as those produced by algae or biological activity, which can cause earthy or musty tastes in drinking water.[10][3]
- Organic chemicals including many pesticides, volatile organic compounds (VOCs), and industrial organics that impact health and flavor.[11][10]
- Chlorine and many chlorine by‑products, including trihalomethanes and haloacetic acids, especially when using catalytic activated carbon.[12][11]
- Some heavy metals and radionuclides in specific conditions, particularly when carbon is specially treated or combined with other media.[13][10]
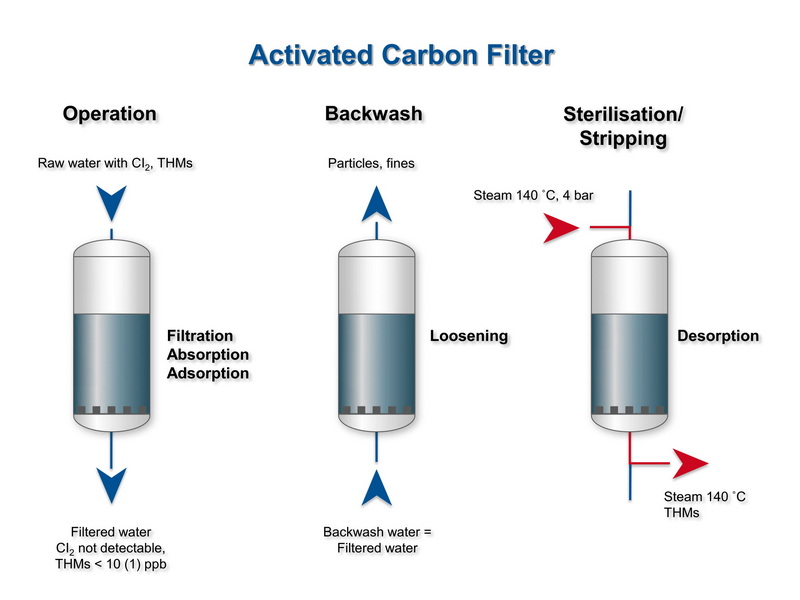
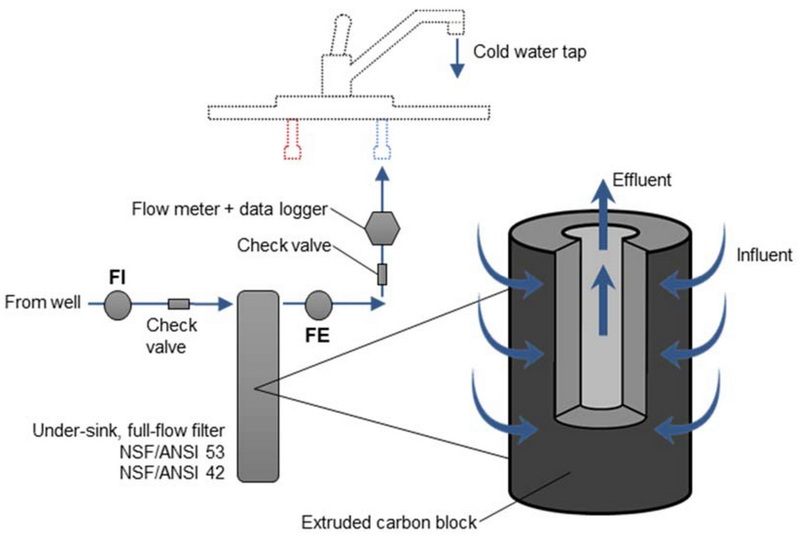
Factors That Control Activated Carbon Performance
The efficiency of activated carbon in water purification depends on both material properties and operating conditions. Understanding these factors helps optimize system design and operation for industrial, municipal, and household applications.[14][4][3]
Key factors include:
- Pore size distribution and surface chemistry of the activated carbon, which determine which molecules can enter and how strongly they are adsorbed.[7][1]
- Contaminant concentration, with higher initial concentrations generally increasing adsorption capacity up to the saturation point.[4][3]
- Contact time and flow rate, where slower flow and longer contact times allow more complete diffusion into pores and better utilization of the carbon bed.[13][3]
- Competing species in water such as natural organic matter, which can occupy adsorption sites and reduce capacity for priority contaminants.[15][3]
In a granular activated carbon bed, water passes through zones where the carbon is fully spent, partially loaded, and fresh, leading to a mass transfer zone that moves through the bed over time. When the mass transfer zone reaches the outlet and contaminants appear at higher levels, this breakthrough indicates that the activated carbon needs replacement or regeneration.[16][17]
Advantages of Activated Carbon in Water Purification
Activated carbon has become a standard technology in drinking water treatment, industrial water polishing, and many point‑of‑use filters because it offers several significant advantages. These advantages make activated carbon an attractive choice for global water projects and product designs.[18][14][3][10]
Main advantages:
- High efficiency for removing taste, odor, and many organic contaminants, including trace organics that pass through many mechanical filters.[13][10]
- Versatility across applications, from large municipal GAC contactors to PAC dosing systems and compact household filter cartridges.[15][3]
- Relatively low operating cost, especially when granular activated carbon is regenerated or reactivated for multiple use cycles.[9][18]
- Compatibility with other processes such as sediment filtration, ion exchange, membrane treatment, and disinfection to build multi‑barrier systems.[14][10]
Limitations of Activated Carbon in Water Treatment
While activated carbon is highly effective for many contaminants, it has clear limitations and should not be viewed as a universal solution. Understanding what activated carbon cannot remove—or does not remove efficiently—is critical for safe and compliant water treatment.[19][14][15]
Major limitations include:
- Poor removal of dissolved inorganic salts and minerals such as hardness (calcium, magnesium), nitrate, and many common ions, which require other processes like ion exchange or reverse osmosis.[19][15]
- Limited effectiveness for microorganisms; activated carbon alone does not reliably remove bacteria and viruses and must be combined with disinfection or fine filtration for microbial safety.[15][13]
- Finite capacity; once the pores of the activated carbon fill with adsorbed contaminants, performance decreases and may even allow previously adsorbed pollutants to desorb if carbon is overused.[17][16]
- No simple visual indicator of exhaustion in most systems, so monitoring or scheduled replacement is needed to avoid breakthrough of contaminants.[9][13]
These limitations mean that activated carbon is often used as part of a treatment train, for example after clarification and filtration but before final disinfection, to ensure robust overall performance.[14][10]
Practical Applications of Activated Carbon Water Purification
In real water purification projects, activated carbon is designed and operated differently depending on scale, contaminant type, and regulatory targets. Both granular activated carbon and powdered activated carbon are widely used in municipal, industrial, and residential systems.[9][3][10][14]
Typical uses include:
- Municipal drinking water plants, where granular activated carbon contactors remove taste‑ and odor‑causing compounds, pesticides, disinfection by‑products, and emerging micropollutants such as certain pharmaceuticals and PFAS.[14][1]
- Industrial process water, where activated carbon protects equipment and products in food and beverage, chemical, and pharmaceutical facilities by removing organic impurities and color.[18][3]
- Sewage and wastewater treatment, where activated carbon filters polish effluents by removing residual organic contaminants and chlorine before discharge or reuse.[18][3]
- Point‑of‑use and point‑of‑entry household filters, where activated carbon cartridges improve taste and odor, reduce chlorine, and remove many common organic pollutants from tap water.[10][11]
In many of these systems, activated carbon is integrated with other technologies such as membranes, UV, or advanced oxidation to meet stricter standards for specific contaminants.[7][14]
Conclusion
Activated carbon works in water purification by adsorbing a wide range of organic contaminants and many taste‑ and odor‑causing compounds onto its extremely large internal surface area, and by catalyzing the reduction of certain disinfectants like chlorine. With carefully designed pore structures, appropriate selection between granular and powdered activated carbon, and proper control of contact time and bed maintenance, activated carbon can deliver high‑quality water in municipal, industrial, and household systems, provided its known limitations for microbes and dissolved inorganic salts are addressed with complementary treatment processes.[4][15][3][14]
FAQ About Activated Carbon in Water Purification
(1) How does activated carbon adsorb contaminants from water?
Activated carbon adsorbs contaminants when dissolved molecules diffuse into its pore system and attach to the carbon surface due to attractive forces such as van der Waals interactions. The vast internal surface area of activated carbon and the dominance of micropores provide many high‑energy sites where organic molecules and other pollutants can accumulate and remain trapped until the carbon is exhausted or regenerated.[2][1][3][7]
(2) What types of contaminants does activated carbon remove best?
Activated carbon removes organic compounds especially well, including many pesticides, VOCs, industrial organics, taste‑ and odor‑causing substances, and numerous disinfection by‑products. It also effectively reduces chlorine and some heavy metals under certain conditions, but its performance is limited for inorganic salts and many dissolved minerals.[15][4][11][10]
(3) What does activated carbon not remove from water?
Activated carbon does not efficiently remove microbial pathogens such as bacteria and viruses, and it also performs poorly on dissolved inorganic ions such as hardness minerals, nitrate, and fluoride. For these contaminants, activated carbon must be combined with disinfection, microfiltration, reverse osmosis, ion exchange, or other specialized technologies to meet quality standards.[19][13][14][15]
(4) How long does activated carbon last in water filters?
The service life of activated carbon depends on contaminant loading, flow rate, and system design, but all activated carbon beds eventually reach saturation and require replacement or regeneration. Because breakthrough can occur before visible changes in water quality, many guidelines recommend scheduled replacement for small filters and performance monitoring or modeling for large granular activated carbon contactors.[17][16][13][9]
(5) What is the difference between granular and powdered activated carbon in water treatment?
Granular activated carbon is used in fixed beds for continuous filtration, allowing longer contact times and often thermal reactivation, while powdered activated carbon is dosed as a fine powder directly into water and later removed by clarification or filtration. In practice, granular activated carbon is preferred for long‑term municipal and industrial systems, and powdered activated carbon is chosen for flexible, short‑term, or emergency treatment where rapid dosing changes are needed.[5][3][4][9]
Citations:
[1](https://www.ncbi.nlm.nih.gov/books/NBK234593/)
[2](https://en.wikipedia.org/wiki/Carbon_filtering)
[3](https://cropaia.com/blog/activated-carbon-in-water-treatment/)
[4](https://www.watertreatmentguide.com/activated_carbon_filtration.htm)
[5](https://www.suezwaterhandbook.com/water-and-generalities/fundamental-physical-chemical-engineering-processes-applicable-to-water-treatment/adsorption/applied-activated-carbon-principles)
[6](https://en.wikipedia.org/wiki/Activated_carbon)
[7](https://ijatec.com/index.php/ijatmmm/article/view/37)
[8](https://generalcarbon.com/understanding-granular-activated-carbon-for-water-treatment/)
[9](https://wqa.org/wp-content/uploads/2022/09/2016_GAC.pdf)
[10](https://extension.purdue.edu/extmedia/WQ/WQ-13.html)
[11](https://www.springwellwater.com/activated-carbon-filters-remove/)
[12](https://www.cleantechwater.co.in/what-do-activated-carbon-filters-remove-from-water/)
[13](https://support.cascadedesigns.com/hc/en-us/articles/35376955864467-What-are-some-of-the-limitations-of-activated-carbon-in-water-treatment-devices)
[14](https://www.epa.gov/sdwa/overview-drinking-water-treatment-technologies)
[15](https://extensionpubs.unl.edu/publication/g1489/na/html/view)
[16](https://www.watertechonline.com/wastewater/article/15549902/the-basics-of-activated-carbon-adsorption)
[17](https://www.karbonous.com/blog/advantages-and-disadvantages-of-activated-carbon-filter/)
[18](https://www.cleantechwater.co.in/blog/what-is-the-use-of-activated-carbon-filter-in-sewage-treatment/)
[19](https://olympianwatertesting.com/exploring-the-advantages-and-limitations-of-activated-carbon-filtration/)
[20](https://greenyplace.com/what-does-activated-carbon-not-remove)
















